Our Projects
|
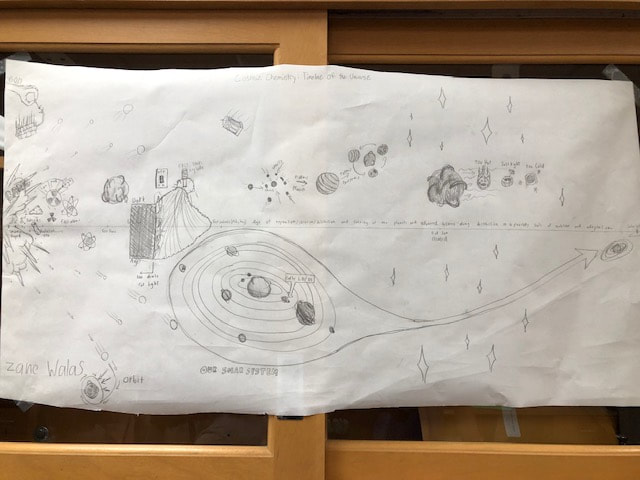
This unit started off with us making a timeline of the big bang to now. We focused on the creation of elements and the phases that matter went through throughout the process. I helped our group research and plan the idea and then did pretty much the entire poster. We then did a full scale version and connected all of the classes research. I helped organize information and then I did artwork for the timeline by request.
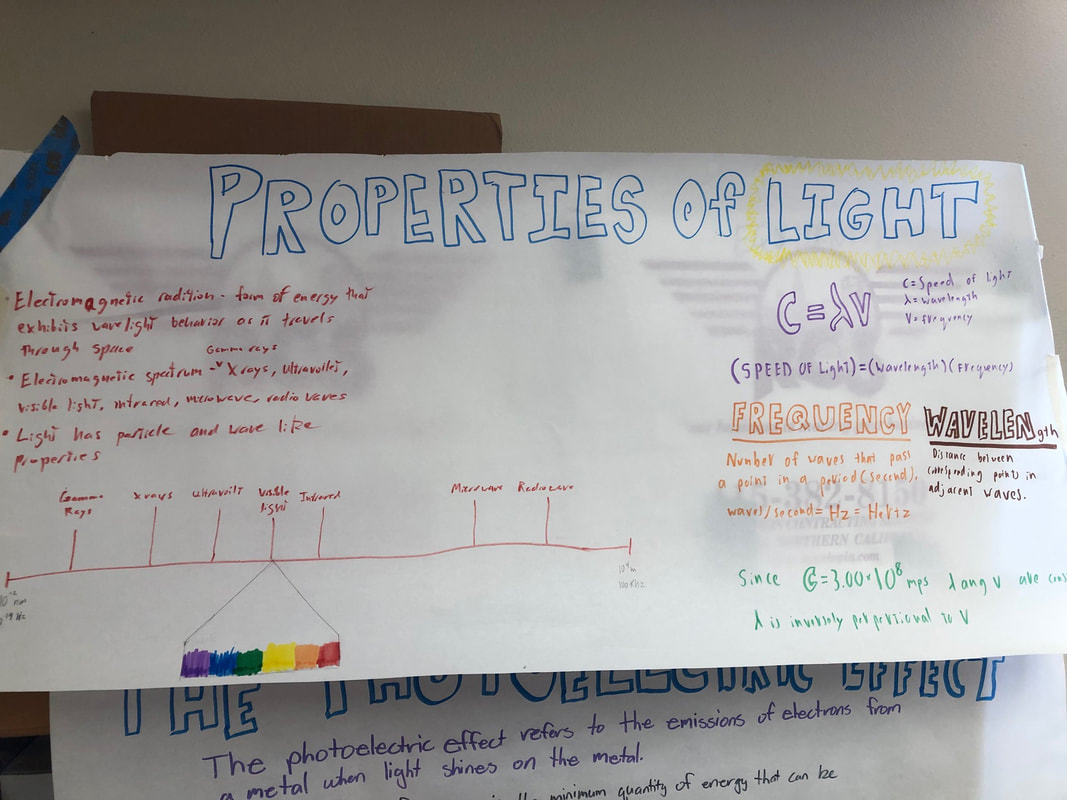
On our second project we read a few pages from the book and had to display the research on a poster. Our topic was the properties of light. I took good notes for the topic and used the notes to help my team choose what to display and how. On our last project we had to research the life cycle of stars and when elements are created by the stars. We created a poster that displayed the stars lives in simple to understand ways and gave them visual characteristics. My teammate with my help researched the elements and wrote about the processes that the stars go through to create them. |
Contents
Proton- A subatomic particle with a positive charge found in the nucleus of an atom. The amount of protons in an atom decides the identity of the element.
Electron- A subatomic particle with a negative charge that is found in the shell and cloud around the nucleus of an atom.
Neutron- A subatomic particle with no charge found inside the nucleus of an atom that accompanies the protons.
Nucleus- Made up of protons and neutrons the nucleus is a tiny ball in the center of an atom.
Isotopes- A radioactive form of an element with a different amount of neutrons.
Ions- An atom or molecule with missing or additional electrons
Elements- Groups of atoms that have a different amount of protons and atomic number. Elements are in their simplest form a single atom and they all have different properties.
Radioactive decay- The process which an unstable nucleus releases rays or a particle to stabilize and release radioactivity.
Alpha decay- When an atom decays a helium nucleus
Beta decay-
Star-
subatomic particles- The particles that make up an atom(neutrons, protons, electrons)
fission- radioactive decay where a nucleus divides into two lighter nuclei.
fusion- When two nuclei combine to create a larger denser nucleus.
half-life- The time in which it takes an unstable element to half its original weight.
nuclear transmutation- The conversion of one element, ion, or isotope into a different element.
radiation- The emission of waves from a radioactive chemical going through radioactive decay.
nucleosynthesis- The synthesis of elements by using smaller nuclei.
Electron- A subatomic particle with a negative charge that is found in the shell and cloud around the nucleus of an atom.
Neutron- A subatomic particle with no charge found inside the nucleus of an atom that accompanies the protons.
Nucleus- Made up of protons and neutrons the nucleus is a tiny ball in the center of an atom.
Isotopes- A radioactive form of an element with a different amount of neutrons.
Ions- An atom or molecule with missing or additional electrons
Elements- Groups of atoms that have a different amount of protons and atomic number. Elements are in their simplest form a single atom and they all have different properties.
Radioactive decay- The process which an unstable nucleus releases rays or a particle to stabilize and release radioactivity.
Alpha decay- When an atom decays a helium nucleus
Beta decay-
Star-
subatomic particles- The particles that make up an atom(neutrons, protons, electrons)
fission- radioactive decay where a nucleus divides into two lighter nuclei.
fusion- When two nuclei combine to create a larger denser nucleus.
half-life- The time in which it takes an unstable element to half its original weight.
nuclear transmutation- The conversion of one element, ion, or isotope into a different element.
radiation- The emission of waves from a radioactive chemical going through radioactive decay.
nucleosynthesis- The synthesis of elements by using smaller nuclei.
Reflection
This project was very fun and I did exceptionally well to lead and actually progress my group through our project. I worked through the poster and stayed on task to get our project to look good. I made the decisions for our group and did well to lead us in the right direction and do more than my share of the work. I did well to diversify my work and make it easy to understand for a broad audience while conveying the ideas and information required. One of my pitfalls was not playing to my teammates strengths as well as mine.I also could have gone for a more difficult or interesting project to display the information in a more clear and effective way. If I were to do this project again I would focus on displaying the information more clearly.