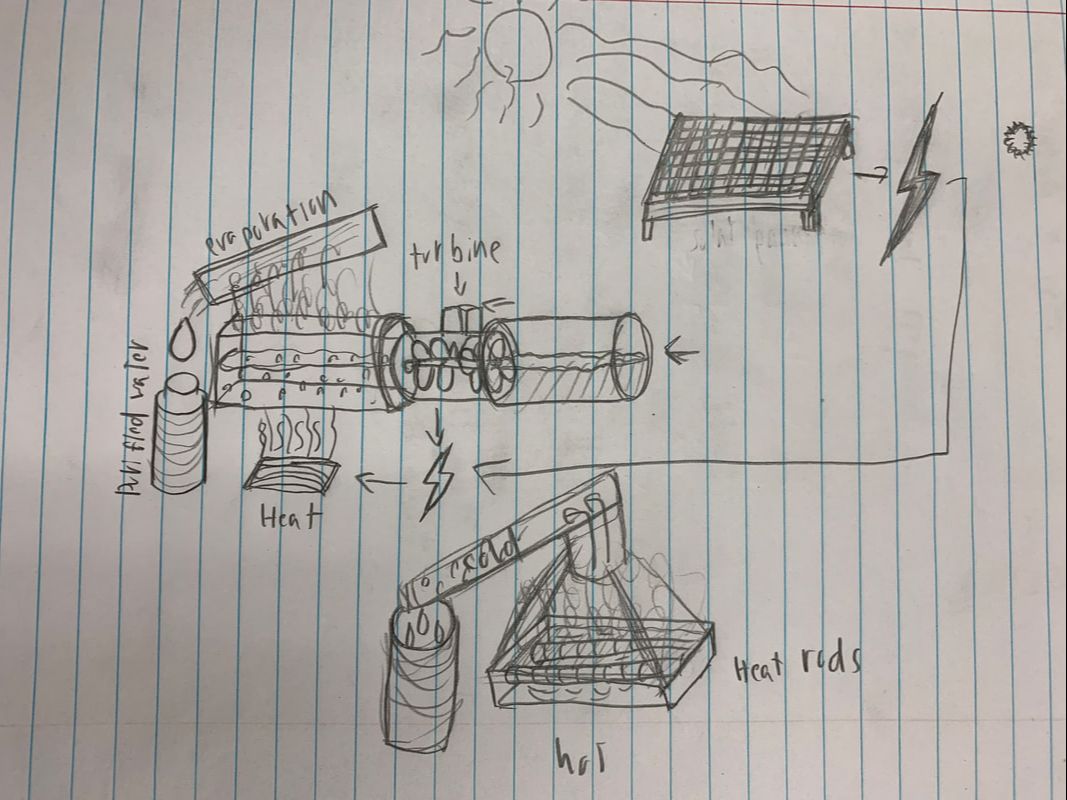
The Electro-Efficient Hydro-Purification Plan
This plan all started with a question. What new age widely available resource can we use to create crystal clear purified water from salt water to still ground water that the president of the water purification company will love? Well here is our answer. This electricity powered purification has revolutionized the water purification game. The plan was thought out collaboratively and I made a sketch of our idea. Electricity is created by turbines and solar power from a water source. Then the electricity is used to heat rods in a metal bath where the unpure water will be stored. When the rods have set the water to boil the steam will rise up to the top and travel towards our cooled tubing where it will condense and the beautiful pure water will trickle down into our purified water cups. Currently water is purified with expensive filters and multiple steps of sterilization and chemical additives. Our plan reduces the need for filters and additives while being a cheap and safer alternative. It kills off all bacteria and through our tests it meets all the regulations drinking water is required to meet.
Contents
|
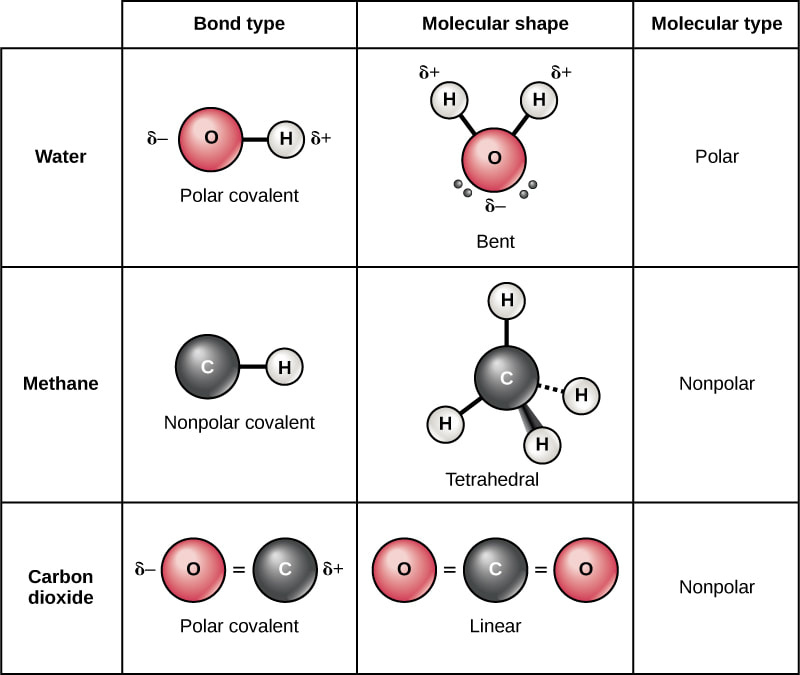
Solution- Chemical substance consisting of two or more elements that were mixed and dissolved into one another.
Solute - Substance dissolved into the solvent Solvent-Substance that dissolves the solute. Often liquid Polarity- Differences in charge that lead to polar bonds in molecules Covalent bond- Chemical bond that involves the sharing of electrons Ionic bonding- Bond between molecules of opposite charge commonly in ionic compounds Polar covalent bond- Chemical bond where the number of electrons is unequally shared. Ph test- Test for the acidity of water. Our ph before was 9 and after was 7. iron test- Test for the levels of iron in water. Our iron before was 2 ppm and after was 0 ppm. chlorine test- Test for the chlorine levels in water. Our chlorine before was zero and after was zero. copper test- Test for the copper levels in water. The copper before was .5 and after was .5. calcium test- Test for the hardness in water. The calcium before was 400 ppm and after was 50 ppm. ppm- Unit of measurement parts per million. |
Reflection
Overall the project went very well. I did a very good job of being hands on and engaged in our groups discussion and work. I also did very well to keep our group from straying to ideas that were not realistic (like waterless showers). I set up my own experiment and gathered data for my group. I also helped plan out and execute our experiments while taking pictures and recording information.